Rollie Swingle didn’t have treatment options for his stage IV prostate cancer.
“The cancer had metastasized into my spine and pelvis,” Swingle says. “Surgery was out. Radiation was out. Hormone therapy (to stop his body from producing testosterone) was the only option.”
Eight years later, he still has cancer, but he’s kept it at bay through faith, his attitude and a willingness to try the latest alternatives.
The American Cancer Society says one in six men will develop prostate cancer, and more than 25,000 new cases will be diagnosed this year in California alone. A quarter of those patients could die from the disease, but new advancements in treatment options and a trend toward more screening — despite controversy — may lower those statistics.
Life on Screen
The standard in screening for prostate cancer — after the infamous glove test — is the prostate-specific antigen (PSA) test. PSA is naturally produced by the prostate gland, so a simple blood test can determine its concentration. Normal levels are usually less than four nanograms per milliliter. Cancer’s likelihood increases as the number approaches 10; Swingle’s was 298.
That said, there is no defined “abnormal” PSA level, and elevated PSA levels do not automatically mean cancer is present.
It’s for that reason PSA tests have been controversial. A study published in the New England Journal of Medicine shows that screening is not saving more lives but is instead causing men to get radical treatment, unnecessarily burdening them with lifelong side effects.
Local experts, however, disagree.
“That study was heavily flawed,” says Dr. David Yee, a urologic oncologist with Sutter Medical Group. “Almost 50 percent of the patients who were not supposed to have the PSA test ended up having one done, diluting the beneficial effect of PSA testing. In reality, prostate cancer deaths have decreased 40 percent since PSA testing started in the early 1990s.”
Dr. Darryl Hunter, a radiation oncologist with Kaiser Permanente and managing partner of the Sacramento Community Cancer Coalition, agrees, saying screening studies don’t accurately account for underserved populations, and the issue of screening really comes down to choices and quality of life.
“If cancer is found early, men have more treatment choices,” he says. “The quantity of life without screening may be the same, statistically, but the quality of life can be much different. Five years in Granite Bay is much better than five in Pelican Bay.”
Today’s Treatment Options
Having more options doesn’t make deciding on a course of treatment easier. No option is perfect, and one option is to do nothing.
“Active surveillance” has become a popular approach for men with elevated PSA levels plus a biopsy showing cancer cells but who are low-risk cases. It involves regular PSA tests and rectal exams, complemented by a schedule of repeat biopsies.
Sometimes an endo-rectal MRI — a procedure in which a screening tool is inserted into the rectum — may be recommended to ensure detection of anything a biopsy may have missed.
The treatment has no side effects. That could lead patients to wonder why they should go through the ordeal of treatment and risk the side effects for something that isn’t yet a threat.
The downside is the cancer can worsen and require more aggressive treatment than may have been initially necessary, begging the question: Why not simply pursue treatment as soon as cancer is found?
“If you follow that thinking, you could say every man with children should have his prostate removed when he turns 50 (to) avoid any cancer risk altogether,” says Dr. Ralph de Vere White, director of the UC Davis Cancer Center. “Our job is to match the appropriate treatment to the diagnosis, not worry patients into something they don’t need.”
When more aggressive treatment is necessary, options include radiation, prostate-removal surgery and hormone therapy. Each has its advocates, and advancements in all offer the promise of better outcomes.
The most aggressive approach is prostate-removal surgery. That can remove the cancer entirely, assuming it hasn’t spread, but comes with the risk of lifelong physical and emotional side effects. The prostate is close to nerves that control bladder and sexual function. That makes surgery tricky; damage to those nerves can lead to incontinence and erectile dysfunction.
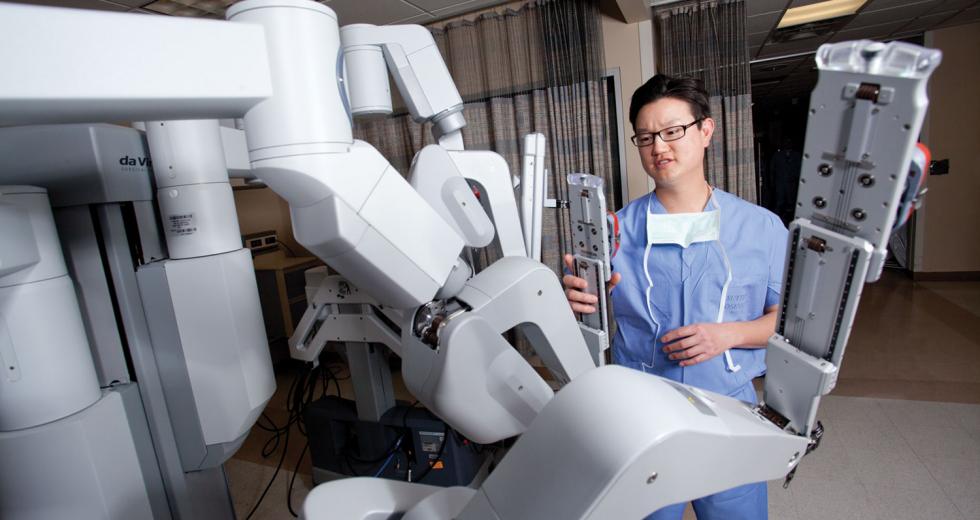
Newer “nerve sparing” procedures minimize these risks, but the surgical option getting the most attention is the robot-assisted approach.
“The da Vinci robot has revolutionized prostate cancer surgery,” Yee says. “Advantages of minimally invasive robotic surgery usually include a shorter hospital stay, less post-operative pain and a faster recovery. Because robotic surgery affords a skilled surgeon with enhanced vision and increased dexterity, there are potentially better outcomes in terms of urinary and sexual function, so it’s not surprising 70 percent of the prostate cancer surgeries in the U.S. today are robotic.”
As an alternative, radiation therapy can be less invasive, and new approaches are enabling doctors to target the cancer much more effectively without damaging surrounding tissues.
There are two forms of radiation therapy. Internal “seed” therapy involves placement of rice-sized radioactive seeds into the prostate. The seeds deliver low-dose radiation over time until they basically burn out.
External beam therapy resembles getting an X-ray. It involves lying on a table while a machine directs high-powered radiation beams into the cancer. Patients typically undergo treatments five days a week for several weeks.
The chronic problem with radiation is that it’s not discriminating — it kills healthy and cancerous cells alike. Given the prostate’s location, this can lead to erectile dysfunction, painful or frequent urination and various rectal symptoms, including bleeding. The prostate also changes position in the body frequently, depending on whether the bladder is full or empty, for example, so the risk of missing the target or hitting too much healthy tissue can be high.
“With radiation, you need an optimal dose distribution,” says Dr. Mark Logsdon, radiation oncologist with Radiological Associates of Sacramento. “The goal is to hit the entire tumor and, to the greatest extent possible, miss everything else. One of the best tools we use to do that is Calypso.”
Calypso GPS technology involves implantation of three nonradioactive seeds into the prostate, which can then be tracked from outside the body. They tell doctors exactly where the prostate is, within one millimeter. Logsdon says even if the prostate moves during treatment, he can turn off the beam, move the patient and start again.
Another option being studied involves injecting a gel between the prostate and the rectum to create a gap of up to 15 millimeters. This space serves as a buffer that blocks much of the radiation before it reaches the rectum.
“We can reduce the radiation dose by half with a distance of just nine millimeters,” Dr. Logsdon says. “We’re looking forward to continuing the clinical studies on this.”
Dr. Logsdon and his colleagues also are preparing a clinical trial with stereotactic ablative body radiotherapy, a high dose of targeted radiation, which could effectively deliver the same amount of radiation currently given over eight weeks in just one week. Those trials are expected to begin this year.
A third option is hormone therapy, although its long-term results can be limited. Simply put, prostate cancer feeds on testosterone. The idea is that by cutting the testosterone supply, the cancer stops growing. What research has learned, however, is that the cancer finds some other way to get what it needs.
Testosterone removal also comes with side effects, ranging from loss of libido and erectile dysfunction to more serious cardiovascular impacts and increased risk of diabetes. Short-term benefits for severe cases, however, are encouraging.
“If you have metastatic prostate cancer, withdrawing its hormonal support is an extraordinarily effective first line of treatment,” says de Vere White. “What we’re finding is we can give it intermittently to lower PSA levels over a period of months, then allow patients to start reproducing testosterone to reverse side effects. Hopefully, 18 to 24 months can pass before PSA levels are elevated again and treatment starts again.”
Hormone treatment certainly worked in Swingle’s case, primarily (in his view) because he was willing to be part of clinical trials. Between 2004 and 2009 he was involved in two trials, both of which had very positive initial results. His PSA levels dropped from 298 to below 50 and then down to eight. Today they hover around 40, which is high but stable.
In 2011, he went beyond traditional therapies and started on the Provenge program, which involves altering a patient’s white blood cells, retraining them to attack cancer.
Other approaches being studied include freezing the cancer cells (cryotherapy) and high-intensity, focused ultrasound. Both are in experimental stages.
Ultimately, the right course for any patient involves numerous variables but more than anything else, it involves what Dr. Logsdon calls the sleep test.
“I tell patients there is more than one right answer,” he says. “Figuring out which one is right for you means finding the approach that is going to let you sleep best at night.”
Recommended For You

Hurts So Good
Yoga is good for you, but be careful boys
I’ve always snickered at yoga.It just seemed ridiculous. But men are flocking to yoga the way we once, in the ’80s, took to this thing called “jogging.” We’re learning that yoga bestows a slew of health benefits — physical, mental, even sexual. But new research also points to increased health risks for men, and this muddies the decision.

Hope for the Iffy Stiffy
Miracle drug or fake science?
Low testosterone. For men, these words have the same foul odor as “impotence,” “shrinkage” or “Justin Bieber.” The topic is taboo. Throughout civilization testosterone has been prized as the lifeblood of manhood, so a deficit would imply, by definition, that we are somehow less manly.