As the world continued to settle into the uneasy stasis of pandemic life in July, Dr. Angela Haczku was already five months into her research on COVID-19.
In March, the respiratory immunologist and associate dean for translational research at the UC Davis School of Medicine was tasked with overseeing the development of research projects related to COVID-19, including the development of testing, therapies and vaccines for SARS-CoV-2, the virus that causes COVID-19. Haczku’s assignment to head up this immense scientific undertaking came just weeks after UC Davis Medical Center had diagnosed the United States’ first case of community-acquired COVID-19 in late February.
“We had a suspicion that the patient had (the virus),” Haczku says, “but testing was not readily available” for patients who didn’t meet the Centers for Disease Control and Prevention criterion at the time that the virus could only be acquired after having direct contact with someone who’d traveled to China, where the virus originated. “Thanks to the insistence and persistence of UC Davis doctors, the CDC and local authorities finally approved our request for testing — and the patient tested positive. Because of our case, the (Food and Drug Administration) relaxed criteria for test development to keep up with the sneaky nature of this disease.”
The local case showed that the virus was no longer localized to China and had spread into the international community, which meant that testing was going to need to be more rapidly and widely available to identify — and, hopefully, help contain — the spread.
The novel coronavirus was first detected in November 2019 and “sequenced” (its genetic makeup determined) by January, so researchers around the world were already working on a vaccine before the general public first became aware of it. The community-acquired case at UC Davis underlined the importance of the work that teams of researchers, faculty members, doctors and other specialists were already doing at the university to create a vaccine for COVID-19. The process, complex in the best of times, was made more urgent by skyrocketing infection rates in early 2020.
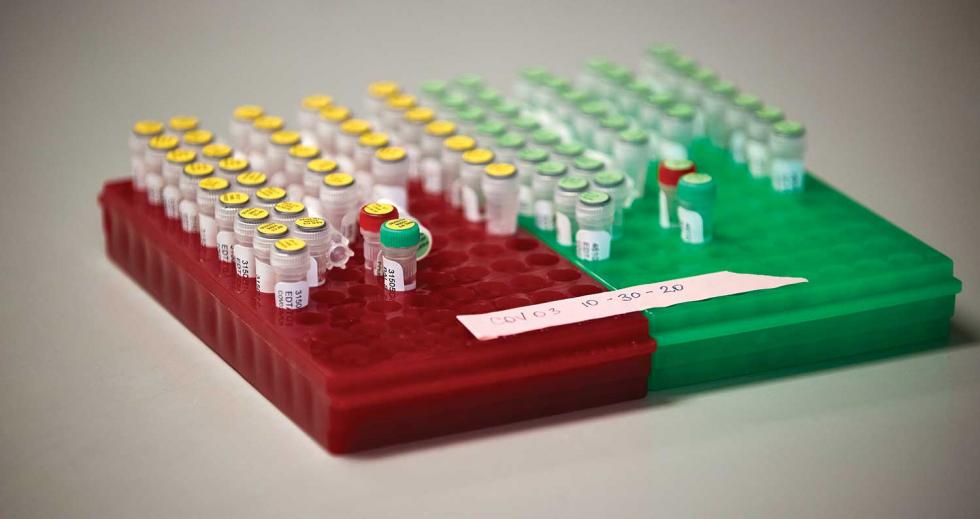
Pedro Hernandez, a UC Davis research associate (left), and Duane
Kim, a UC Davis medical student, conduct an experiment in Dr.
Angela Haczku’s lab to stimulate airway epithelial cells, which
protect the body against pathogens and are likely targeted by
COVID-19. (Photos by Fred Greaves)

Speed is certainly a necessity but so too are accuracy, safety and efficacy, and it’s up to the experts around the world to figure out how to balance the equation. The efforts of the UC Davis teams and those of the international science community have led to UC Davis’ participation in a global clinical trial being run by Pfizer — one of the most promising vaccine trials to date. In fact, Pfizer and partner company BioNTech announced Nov. 9 that their vaccine candidate had proven 90 percent effective at preventing COVID-19 in trial participants who had no prior evidence of infection.
The Making of a Vaccine
Over the centuries, vaccines have become a critical piece of health care to protect populations from virulent diseases and, sometimes, even eradicate them. Their use can be traced back as far as 10th-century China, though the term “vaccine” wasn’t used until the late 1790s when English scientist Edward Jenner published his work with variola vaccinia (cowpox), which led to the first vaccine for human smallpox.
Though there are many types of vaccines, their fundamental mechanism is the same. By introducing a foreign agent into the body, a vaccine stimulates the immune system to recognize the threat and mount an attack, creating antibodies (also known as immunoglobulins) that destroy the intruder and remember it in case it returns. If a person is later infected with an actual pathogen, like a bacterium or virus, the body recognizes it from before and neutralizes the threat before the intruder can infect the body’s cells.
If it’s already infected some of the cells, the immune system destroys those cells to keep the agent from multiplying. Haczku explains that the responsiveness of the immune system to a vaccine is measured by the number of antibodies the body is able to mount in response to the injection, which, in turn, determines the effectiveness of the vaccine.
Dr. Dennis Hartigan-O’Connor, an associate professor in the UC
Davis Department of Medical Microbiology and Immunology, studies
a COVID-19 vaccine candidate in his lab.

Once a vaccine has been developed, it must go through a rigorous testing process. In the preclinical stage, a vaccine is tested for efficacy on animal models (test subjects) before moving on to human subjects. Mice, rats and nonhuman primates are the most used species for testing COVID-19 vaccines, since rodents can be genetically altered to be susceptible to the virus, and primates — specifically rhesus macaque monkeys — most closely resemble humans in both their immune response and development of symptoms when infected with SARS-CoV-2.
In August, the National Institutes of Health awarded the UC Davis Mouse Biology Program a $1.2 million grant to create mice that are susceptible to COVID-19 (they aren’t naturally) and distribute them to other researchers. On the primate side, rhesus macaque testing for COVID-19 has been underway since March at the on-campus California National Primate Research Center, one of seven such centers across the country funded by the NIH developed to breed, house, care for and study primates for medical and behavioral research. What distinguishes UC Davis’ research is the CNPRC’s unique collection of primates at various stages of health and age, a key factor in making sure a vaccine will work to protect the breadth of the human population, including those with underlying health conditions.
“Animal models are extremely important, because if we can provoke a classic immune response in an animal, we can use that information to develop a vaccine,” explains Dr. Smita Iyer, an immunologist and assistant professor at the CNPRC, the UC Davis School of Veterinary Medicine, and the Center for Immunology and Infectious Diseases. Iyer is studying how helper T cells (a type of immune cell that activates the body’s immune response) work against SARS-CoV-2. “Human studies are harder to control depending on when you get samples and how much you get. Those ambiguities are removed in an animal study” because of the more controllable scientific environment.
“Animal models are extremely important, because if we can provoke a classic immune response in an animal, we can use that information to develop a vaccine. Human studies are harder to control depending on when you get samples and how much you get. Those ambiguities are removed in an animal study.”
DR. SMITA IYER Immunologist and assistant professor UC Davis School of Veterinary Medicine
After animal testing, a vaccine enters the clinical development stage, a three-phase process defined by the CDC as Phase I: “small groups of people receive the trial vaccine”; Phase II: “vaccine is given to people who have characteristics (such as age and physical health) similar to those for whom the new vaccine is intended”; and Phase III: “the vaccine is given to thousands of people and tested for efficacy and safety.” (Even after being approved and licensed by the FDA, many vaccines continue to be monitored by the agency, which is considered Phase IV.) All told, the development of a vaccine from lab to public consumption can take up to a decade or more, with up to four years being spent in the preclinical stage, then two or more years in human clinical trials, then in continued assessment after a vaccine has been approved, licensed, manufactured and widely distributed.
Types of Vaccines
UC Davis’ researchers have been studying three classes of vaccine to combat SARS-CoV-2: DNA, mRNA and protein vaccines.
Vials in the lab of Dr. Smita Iyer, an immunologist and assistant
professor at the UC Davis School of Veterinary Medicine, hold
tissue samples for her COVID-19 vaccine research.
In DNA, or “gene therapy,” vaccines, researchers inject genetic material from an antigen (the molecule present on the outside of a pathogen) into a patient, causing a small number of the patient’s cells to replicate the material, which, in turn, stimulates the patient’s immune system to fight off the antigen.
In mRNA vaccines, scientists introduce a nucleic acid sequence that mimics a specific antigen, which prepares the body to recognize and fight it if infected with the real thing. Of the hundreds of DNA and mRNA COVID-19 vaccine trials Haczku says are in progress worldwide, only about 10-15 will lead to human trials.
In protein vaccines, a patient is injected with a solution that contains the deactivated protein of an actual pathogen, which causes the body to mount an immune response — and later remember and destroy that same protein if it’s encountered again. This type of vaccine is the most commonly used and simplest to make.
DNA vaccines are a specialty of Dr. Dennis Hartigan-O’Connor, an associate professor in the UC Davis Department of Medical Microbiology and Immunology and a core scientist at the CNPRC. His research on the immune system and chronic diseases like AIDS and his work with primates has brought him international recognition and a strategic spot on the team for developing a vaccine for the novel coronavirus.
“Our work at UCD is two-pronged. We’re contributing to the nationwide effort to evaluate (COVID-19) vaccines and therapies in animal models and also bring forward candidates that could bring herd immunity and long-term coverage. With our long history of testing HIV vaccines at UCD, we’re adapting those candidates and using them as a foundation.”
DR. DENNIS HARTIGAN-O’CONNOR Associate professor UC Davis Department of Medical Microbiology and Immunology
“Our work at UCD is two-pronged,” says Hartigan-O’Connor, who recently licensed one of the vaccine candidates developed in his lab to startup company Tendel Therapies, which will work to get it evaluated by the FDA and then hopefully into Phase I trials by February 2021. “We’re contributing to the nationwide effort to evaluate (COVID-19) vaccines and therapies in animal models and also bring forward candidates that could bring herd immunity and long-term coverage. With our long history of testing HIV vaccines at UCD, we’re adapting those candidates and using them as a foundation. There’s a tremendous amount of data that is very encouraging that these vaccines could elicit a response (against COVID-19).”
Operation Warp Speed
Though he’s optimistic about the progress being made at UC Davis, Hartigan-O’Connor is well aware that the accelerated demand for an effective vaccine has complicated an already difficult process. During the highly politicized race for a vaccine before the November U.S. presidential election, scientists warned that not only was a vaccine unlikely in that short time frame, but also that anything rushed through approvals might not be safe.
Dr. Angela Haczku, a respiratory immunologist and associate dean
for translational research at the UC Davis School of Medicine,
oversees the university’s development of research projects
related to COVID-19.

That left researchers like Haczku, Iyer and Hartigan-O’Connor to not only work around the clock as part of Operation Warp Speed — the White House initiative launched in May to “accelerate the testing, supply, development, and distribution of safe and effective vaccines, therapeutics, and diagnostics to counter COVID-19 by January 2021” — but also to manage expectations.
“We have plenty of tools to design a vaccine, but on a time scale that’s frustrating to people,” says Hartigan-O’Connor. “The process of sorting through those tools is a lengthy one. There are two pieces to a (successful) vaccine: It has to create an immune response, and the response has to be effective. We know we can create the response, but we don’t know if that will be enough. We have to follow (test subjects) for a minimum of two years to rule out deleterious effects and see how long the vaccine lasts.”
Hartigan-O’Connor says that many of the vaccines currently being tested in humans and nearing final approval by the FDA may work, but that they’re a “simpler” form of vaccine, meaning they rely on less sophisticated mechanisms (like an inactivated virus) and therefore don’t provide long-term protection without booster shots. Though more highly optimized vaccines take longer to develop due to their more complex design, they also often provoke a more intense immune response that lasts longer.
One of the most promising vaccine candidates already in human clinical trials is being tested in part at UC Davis. Aug. 12, UC Davis Health announced that it had entered into a partnership with Pfizer and BioNTech, a German biotechnology company whose lead mRNA vaccine researcher is a longtime colleague of Haczku’s, to participate in a 30,000-person global study. UC Davis was one of 120 sites across the country and one of two in the Sacramento area (along with Kaiser Permanente) selected for the trial due to its ethnically diverse population and high prevalence of infection. Approximately 200 people are participating in the UC Davis study.
Dr. Dennis Hartigan-O’Connor, an associate professor in the UC
Davis Department of Medical Microbiology and Immunology, recently
licensed one of the vaccine candidates developed in his lab to
startup company Tendel Therapies.

“This is a huge undertaking, even though we’re only a very small cog in the machinery,” says Haczku, who started administering first-round injections Aug. 20 and finished at the end of September. “An mRNA vaccine (like this one) has not been used in human history to combat infectious disease. It’s (a) historic first.”
Following this first round of injections, participants returned to UC Davis after 21 days for a booster, which will then be followed by six-month, one-year and two-year follow-ups to track how long the immunity lasts and if the vaccine has actually prevented anyone from getting sick. Haczku explains that the analysis of the study results is performed by the Data Safety and Monitoring Board, an independent body appointed for the clinical trial. (All investigators, like those at UC Davis, are blinded in a study of this kind, meaning they don’t know whether their participants received the actual vaccine or a placebo. The DSMB, conversely, is unblinded and can therefore analyze interim results.)
Based on the first interim efficacy analysis of the trial conducted Nov. 8, the DSMB found that there have only been 94 COVID-19 positives so far out of the more than 45,000 participants who eventually received the vaccine. Comparing these results to those of placebo recipients, they found that the vaccine was 90 percent effective in preventing the disease in participants who had not already contracted COVID-19. Haczku anticipates that based on these results, the FDA will approve the vaccine and an emergency-use authorization will soon be issued. Upon approval, UC Davis will likely be one of the distribution hubs because of its availability of freezer space (the vaccine must be stored in specialized freezers that can reach minus 80 degrees to maintain efficacy).
Dr. Smita Iyer, an immunologist and assistant professor at the UC
Davis School of Veterinary Medicine, is studying how helper T
cells (a type of immune cell that activates the body’s immune
response) work against SARS-CoV-2.

While the Pfizer study continues to demand time and attention, Haczku and her UC Davis colleagues are still working on other relevant research. Haczku is the principal investigator on a study that’s been going since April testing the VaxiPatch, a vaccine delivery system designed by biopharmaceutical company Verndari that could eventually be used for COVID-19. And at the beginning of October, Haczku and Hartigan-O’Connor began collaborating on a study examining the role an innate immune molecule might play in fighting the novel coronavirus.
“As we’re working toward a long-lasting and effective vaccine at UCD and elsewhere, people need to use every tool they have,” Hartigan-O’Connor says. “We’re going to have a vaccine, but those first candidates will need help from social distancing and masks for many months into the future, if not years. All of these different measures will need to work together.”
–
Stay up to date on the coronavirus and its effects on business in the Capital Region: Subscribe to the Comstock’s newsletter today.
Recommended For You
The Image of Health
Co-inventor Simon Cherry on how the world’s first total-body PET scanner could significantly change disease diagnosis
Comstock’s spoke with Cherry about the groundbreaking work that led UC Davis to name him and co-inventor Ramsey Badawi among the recipients of the 2020 Chancellor’s Innovation Awards.

Status Check: Health Care Hanging On
Local leaders say they’re undaunted by intense fiscal impacts from COVID-19
COVID-19 has presented significant challenges for health care. At least for the moment, though, local providers have been hanging tough and looking toward economic recovery.

Here’s What Happens to Science When California’s Researchers Shelter in Place
After California Gov. Gavin Newsom’s order for Californians to shelter in place, university campuses across the state began gauging the risk of letting research continue. And scientists are finding their work, plans and lives upended by the spread of the novel coronavirus.

Clear Vision for the Future
VSP Global President and CEO Michael Guyette on overseeing the nation’s only not-for-profit vision benefits company
Michael Guyette became president and CEO of VSP Global, based in Rancho Cordova, in 2018. Comstock’s spoke to Guyette about leading the 65-year-old organization.




